Medical Devices DECODED
Previous edition: 09 May 2024
Share article
Get the full version straight to your inbox.
Exclusive access to our best-in-class data & intelligence
Subscribe now
Qiagen and FBI partner on DNA quant digital PCR assays
The collaboration aims to develop a first-of-its-kind digital PCR assay capable of detecting and quantifying minimal amounts of DNA with high accuracy.
The US Federal Bureau of Investigation (FBI) is partnering with Netherland-based Qiagen to develop a novel assay capable of detecting and quantifying minimal amounts of DNA with high accuracy.
The first-of-its-kind digital polymerase chain reaction (PCR) is expected to boost forensic DNA evaluation to create a better DNA profile.
Forensic samples usually contain a small amount of DNA, PCR can be useful for expanding the sample for evaluation. Additionally, the DNA sample can be damaged by age or environmental factors such as soil. Digital PCR has a higher tolerance to these inhibitors compared to traditional PCR and allows for higher accuracy, according to Qiagen.
Qiagen has previously worked with the FBI, on next-generation sequencing (NGS) technology, ForenSeq MainstAY workflow, to improve workflow. The technology allows for standardised workflow while allowing for more samples to be evaluated, up to 96 samples per run. Qiagen added ForenSeq MainstAY workflow to its portfolio as part of the $150m acquisition of Verogen, in January 2023.
The new digital PCR assay will be run on Qiagen’s QIAcuity digital PCR devices. The QIAcuity platform is an automated nanoplate-based system that counts the presence or absence of DNA molecules. It generates results in approximately two hours.
The market for PCR systems is expected to grow from being worth approximately $700m in 2023 to over $1.2bn in 2033, as per GlobalData market analysis. The digital PCR system segment is expected to see growth of over $300m, with the market growing from being worth $480m in 2023 to $815m in 2033.
Apart from forensics, PCR systems are commonly used as an in vitro diagnostic for the detection of different viruses. In February, Autonomous Medical Devices Incorporated (AMDI) received a National Institutes of Health (NIH) Rapid Acceleration of Diagnostics (RADx) Tech award of up to $5.3m in financing to develop a fast PCR system. The system is expected to detect up to 32 targets per sample in less than ten minutes.
Latest news

15th Annual Outsourcing in Clinical Trials East Coast 2024
The upcoming Outsourcing in Clinical Trials Europe 2024 conference will explore advancements in the clinical trial industry.

FDA issues warning on Getinge device amid quality concerns
Following a series of voluntary recalls, the FDA has urged healthcare providers to avoid Getinge’s intra-aortic balloon pumps over safety concerns.

4C Medical's AltaValve gains dual breakthrough designations from FDA
The device has received the designation for treating mitral regurgitation and mitral annular calcification.

UK report warns that economic instability is threatening life sciences sector
The report published by the Medicines Discovery Catapult warns that a mix of economic uncertainty and lack of facility access is pushing investors away from Britain’s healthcare SMEs.
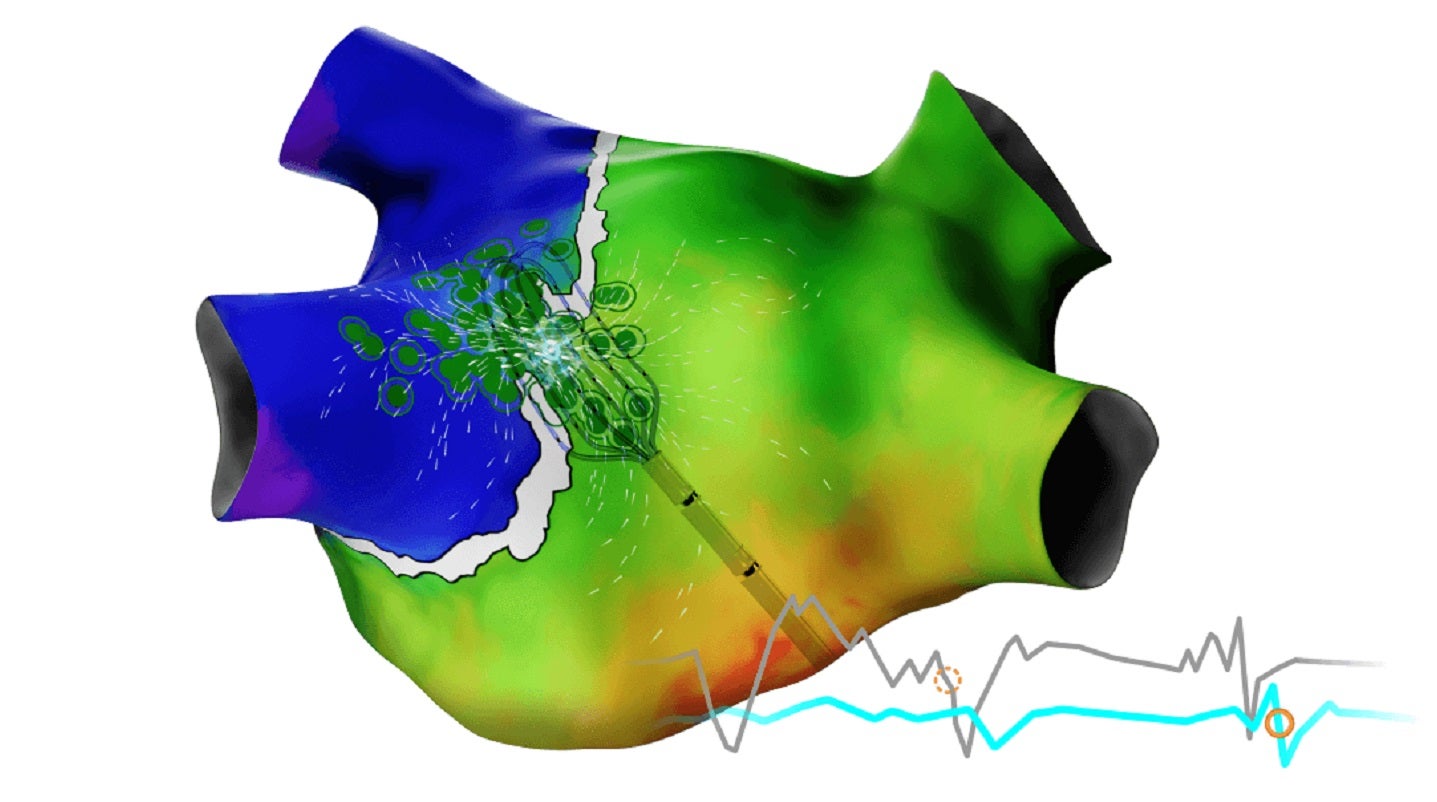
Biosense introduces CARTO 3 System Version 8
The latest version features new modules, including the CARTO ELEVATE Module and CARTOSOUND FAM Module.

Vivalink introduces new solution for MCT and Holter monitoring
The new technology combines RPM technologies with advanced arrhythmia detection algorithms.
iHealth's COVID-19/Flu rapid test secures FDA EUA
Intended for over-the-counter use, the 3-in-1 test is intended for detecting Covid-19 and influenzas A and B.

Biotechs must prioritise ethics to deter GenAI “bad actors”
At the London Biotechnology Show, Microsoft’s senior director of life sciences discussed the ethical use of GenAI.

Leica Microsystems adds Viventis Microscopy to portfolio
The addition of Viventis adds light sheet technology to Leica’s line of research microscopes.
In our previous edition

Medical Devices Decoded
Samsung to acquire foetal ultrasound AI software company Sonio
08 May 2024

Medical Devices Decoded
Medtronic's renal denervation system receives approval in China
07 May 2024

Medical Devices Decoded
Angle signs AstraZeneca deal to develop prostate cancer assay
06 May 2024
Newsletters in other sectors
Aerospace, Defence & Security
Medical Devices
Travel and Tourism
Search companies, themes, reports, as well as actionable data & insights spanning 22 global industries
Access more premium companies when you subscribe to Explorer