Pharma DECODED
Previous edition: 09 May 2024
Share article
Get the full version straight to your inbox.
Exclusive access to our best-in-class data & intelligence
Subscribe now
Register Now
Now Software Inc has extended it time and contact management software, Now Up-to-date, to run under Windows. The Portland, Oregon-based company said the move would expand its corporate customer base. Now Software sells mainly to large corporates that use a mixed environment of Macintosh and Windows personal computers and therefore need software than will run under both operating systems. Now Up-to-Date includes QuickAccess Manager, a permanent on-screen toolbar that gives users access to schedules, name and numbers, and additional diary dates that when entered in the QuickPad automatically appear under the appropriate date. The company said that its main rival was Microsoft Corp's Schedule Plus, which is given away with Microsoft Office. Now said that it had the edge over Schedule Plus because Now Up-to-date ran under more operating systems. It claims to be compatible with Macintosh, Windows 3.1 and Windows95. Now said it is currently working on expanding its software so it can be accessed by remote users with Personal Digital Assistants and pagers. It is also looking at making the software available on corporate Web servers so that companies can offer this information as part of their Web sites. The single-user version costs #100 and will be available this week. The workgroup edition for five users will costs #475, a 10-user version will costs #875 and a 50-user version will cost #3,900. Workgroup editions will be shipping next month.
Latest news
ESCMID 2024: Emerging bacterial infections in neonates and children
Coinfections can complicate disease management and hence continued surveillance and targeted intervention strategies are needed.

FDA fast track status granted to iECURE neonatal OTC deficiency treatment
ECUR-506 is being evaluated in the first-in-human Phase I/II OTC-HOPE study.
Sobi's Aspaveli obtains indication extension approval in Europe
The approval for Aspaveli's extended indication is based on data from the APL2-308 (PRINCE) study.

Biotechs must prioritise ethics to deter GenAI “bad actors”
At the London Biotechnology Show, Microsoft’s senior director of life sciences discussed the ethical use of GenAI.

Zenas raises $200m to advance autoimmune antibody therapy
Zenas signed a licensing deal for Asia-Pacific territories for the bispecific antibody therapy, obexelimab, with Bristol Myers Squibb in September 2023.

Regenex Bio eyes FDA meeting to confirm pivotal DMD trial
Regenex Bio will sit down with the FDA in Q3 2024 to finalise the Phase III trial design.
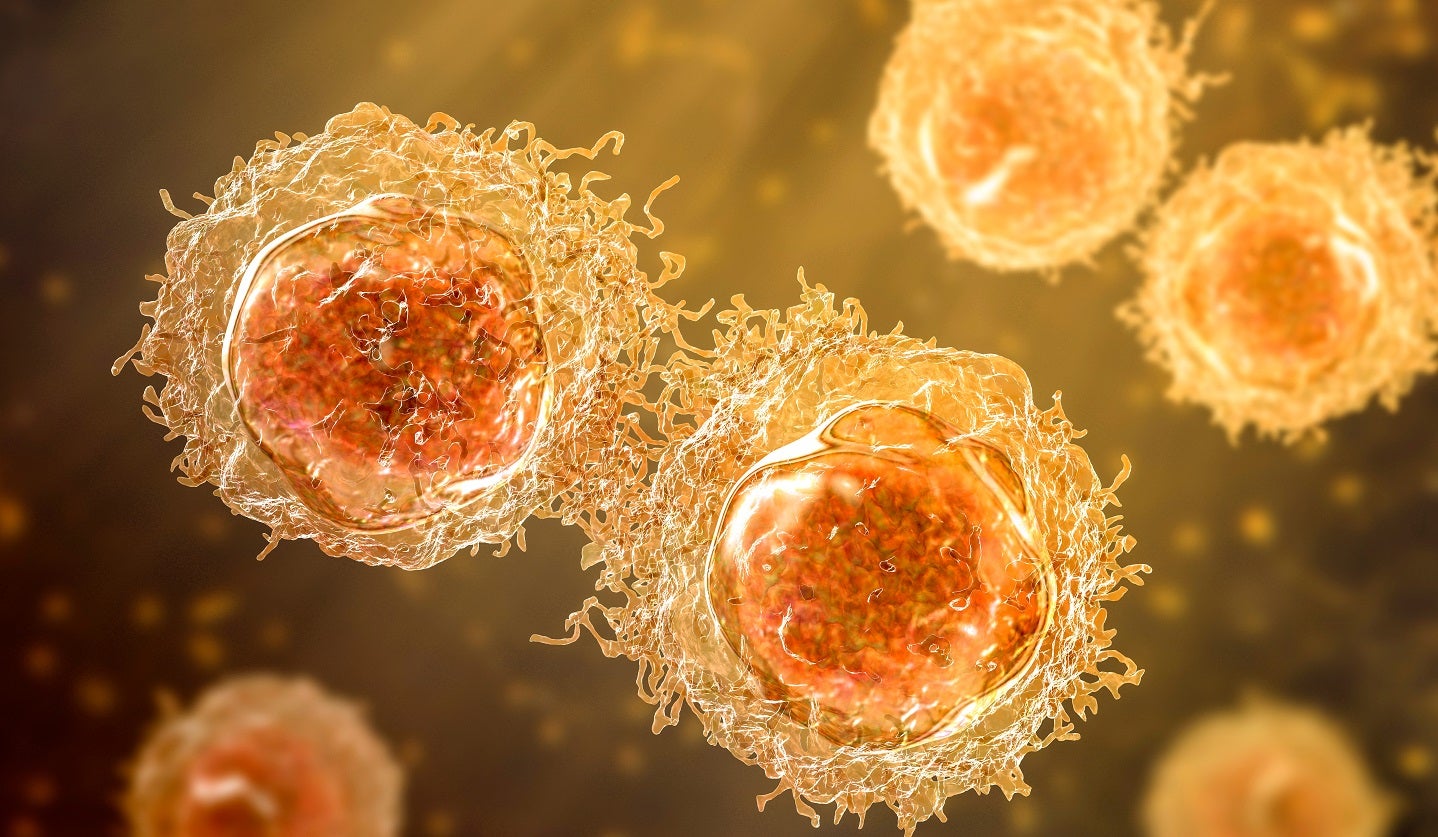
Acepodia reports preliminary results from ACE1831 Phase I trial
The preliminary data indicates potential benefits for non-Hodgkin's lymphoma patients.

Teva and Medincell report positive data for TEV-‘749 in schizophrenia trial
TEV-‘749 has met primary endpoint in Phase III trial, showing significant symptom reduction in schizophrenia patients.

15th Annual Outsourcing in Clinical Trials East Coast 2024
The upcoming Outsourcing in Clinical Trials Europe 2024 conference will explore advancements in the clinical trial industry.
In our previous edition

Pharma Decoded
AstraZeneca to withdraw Covid-19 vaccine globally
08 May 2024

Pharma Decoded
BioNTech reports net loss of €315.1m in Q1 2024
07 May 2024

Pharma Decoded
Amgen reports a 22% increase in Q1 2024 net revenues
06 May 2024
Newsletters in other sectors
Aerospace, Defence & Security
Medical Devices
Travel and Tourism
Search companies, themes, reports, as well as actionable data & insights spanning 22 global industries
Access more premium companies when you subscribe to Explorer